With no new classes of antibiotics for decades and resistance to all antimicrobial drugs on the rise, an urgent search is under way to develop innovative new biological alternatives, explains Craig Billington of ESR
The growing concern around the world about the emergence of bacterial strains showing resistance to all classes of antibiotics, highlights the urgent need to find alternatives to what have been life-saving drugs.
No new classes of antibiotics have been developed for 30 years and resistance has arisen to all antimicrobial drugs developed to date.
Alarmingly, there is also an increasing prevalence of multidrug-resistant bacterial strains internationally, rendering some patients in effect untreatable. To address this, the World Health Organisation, the Food and Agriculture Organisation and the World Organisation for Animal Health have jointly called for urgent action to develop new antimicrobial agents as alternatives to antibiotics for use in agricultural production and clinical care.
At present, these multidrug-resistant bacterial strains are not yet well established in New Zealand, so now is the time to act on introducing new treatments.
ESR scientists are part of a team including experts from the University of Canterbury looking at developing innovative new biological alternatives to conventional antibiotics.
This has the potential to save lives, reduce healthcare costs, ensure the sustainability of New Zealand’s agricultural food production and protect our export driven economy.
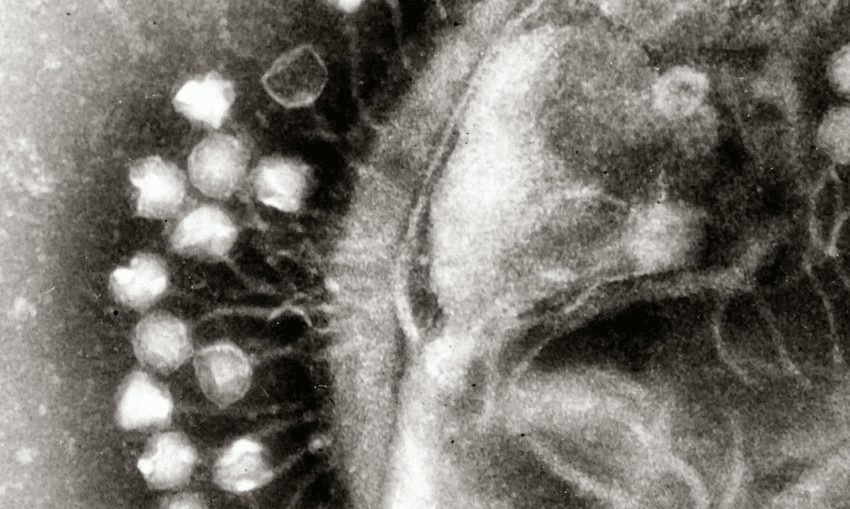
The work will lead to a new class of alternative treatments – based upon innovative bacteria-killing enzymes that are likely to be well tolerated by both humans and farm animals.
The enzymes are naturally generated by bacteriophages which are viruses that only infect bacteria, during their infection cycle.
There is also evidence that the development of resistance to such enzymes may be considerably more difficult compared with conventional antibiotics treatments due to their targeting of critical cell wall components which are highly evolutionarily conserved.
In addition, the very specific nature of the interaction of the phage enzymes and bacteria means that the target organism can be attacked while other organisms remain unharmed. This would enable beneficial bacteria, for instance in the gut, to survive reducing some of the side effects of conventional antibiotic treatment. Unlike conventional antibiotics, the enzymes will naturally biodegrade and so will not persist in the environment, enabling more sustainable agricultural practice.
The potential for using phages to kill bacteria has been recognised for almost 100 years, with early pioneering work being carried out at around the time of the First World War.
Despite early promise, phage therapy of human disease was not adopted in western countries and eventually the development of antibiotics in the 1920s ended the initial era of phage therapeutics, although the use of phages to treat infection was used extensively in the former Soviet Union throughout the 20th century, not least because antibiotics that were widely adopted across the western world were difficult to access in Soviet states.
For all that, phages were used in the west. Indeed, there are reports that phage therapy enjoyed a moment of Hollywood glamour in 1961, with the then film star Elizabeth Taylor hitting the headlines when she was treated for pneumonia with the use of a staphylococcus bacteriophage. We are now looking to exploit the way these bacteriophages kill bacteria by harnessing the power of their enzymes.